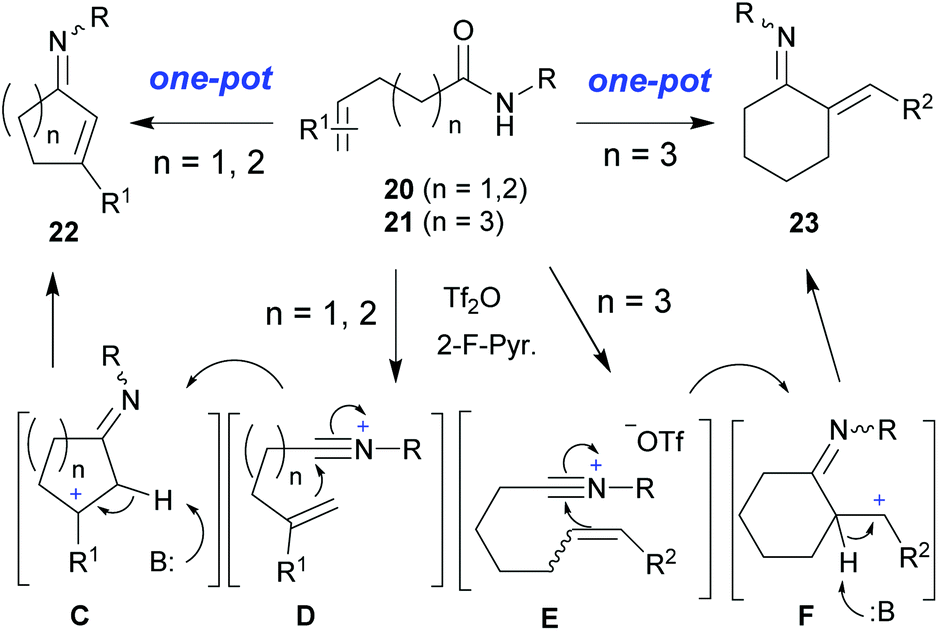
One-pot synthesis of N-heterocycles and enimino carbocycles by tandem dehydrative coupling–reductive cyclization of halo- sec -amides and dehydrative ... - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C6QO00720A
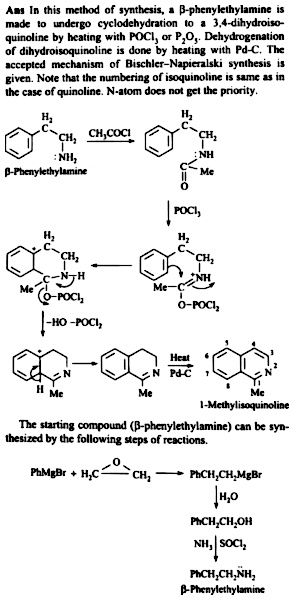
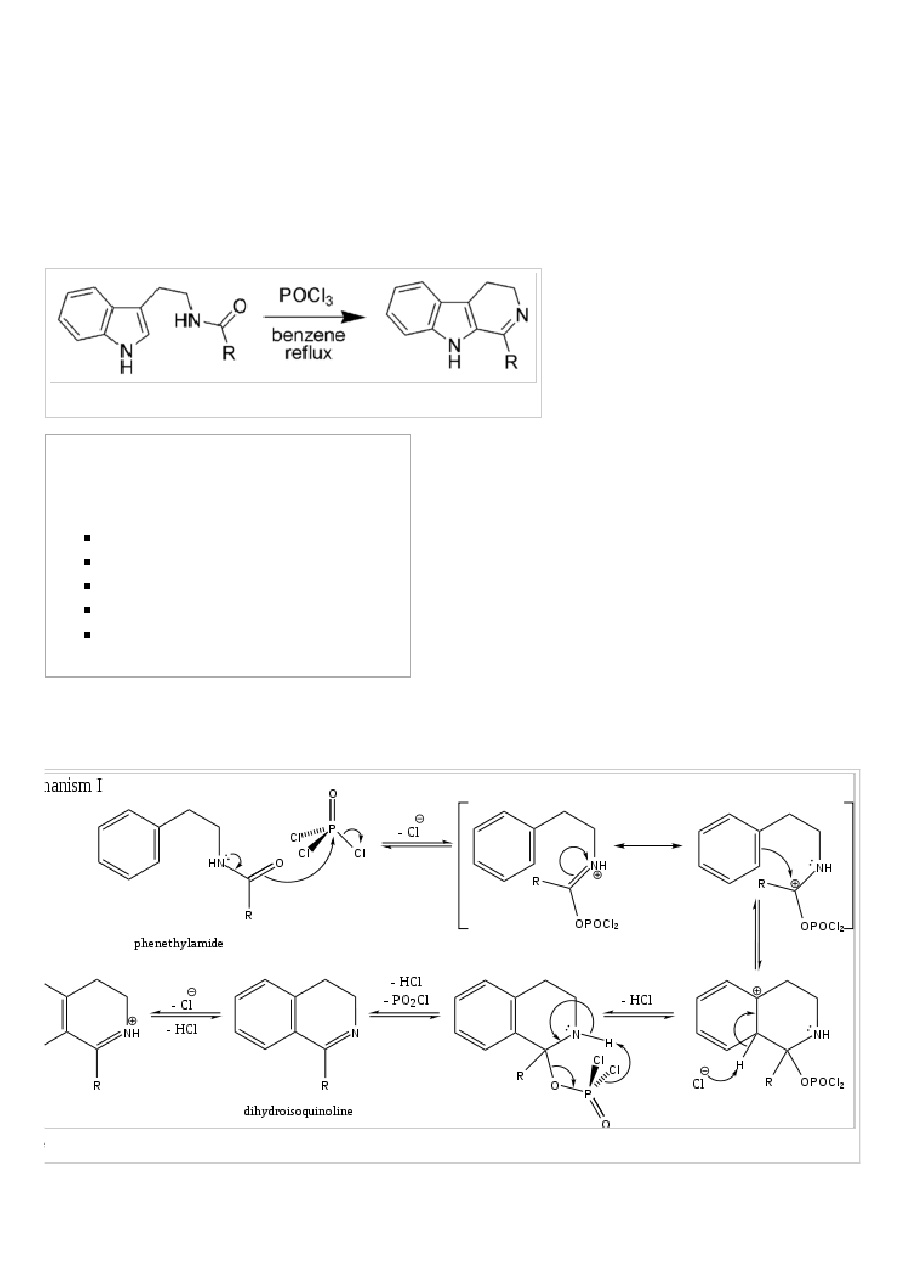
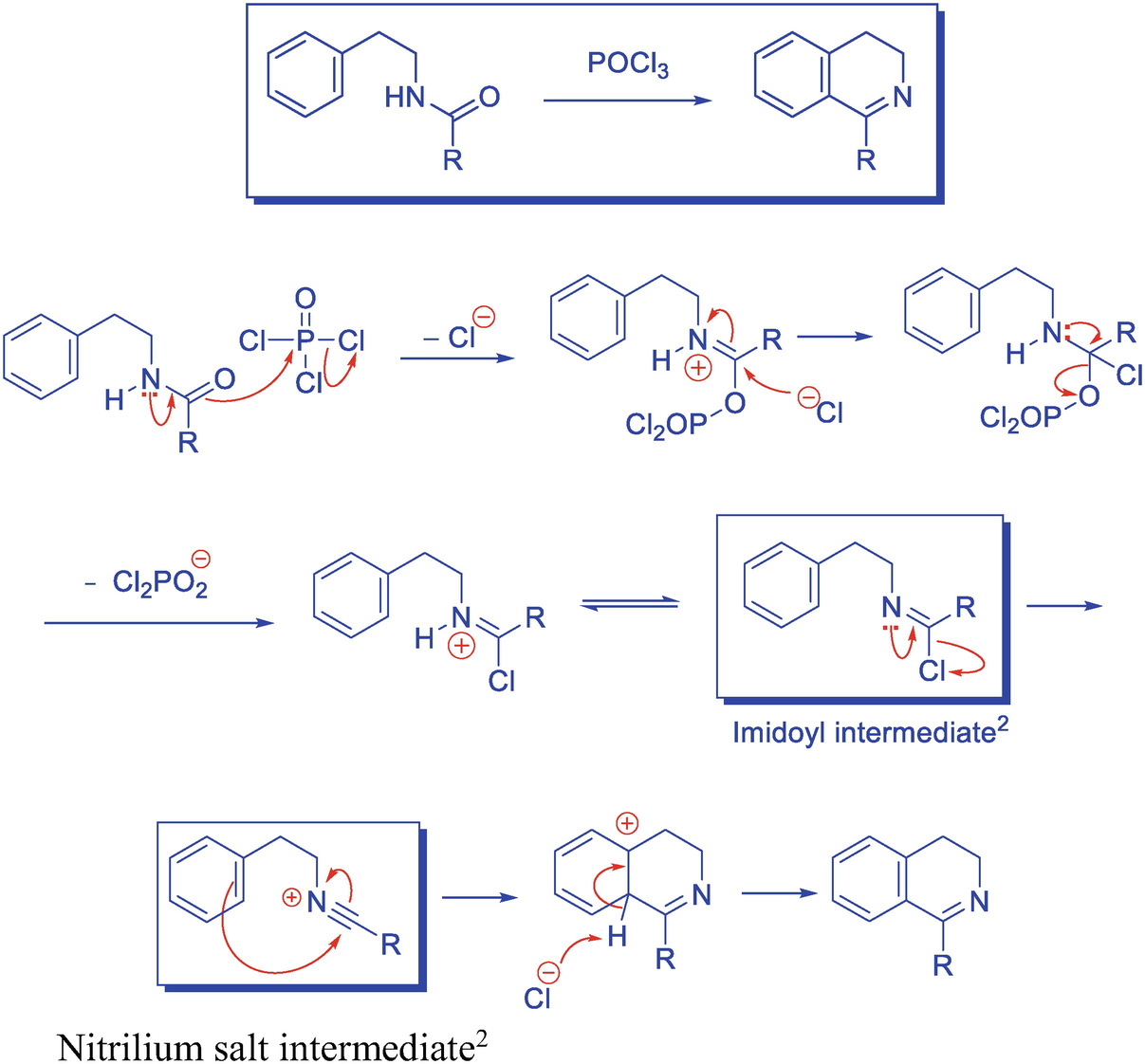
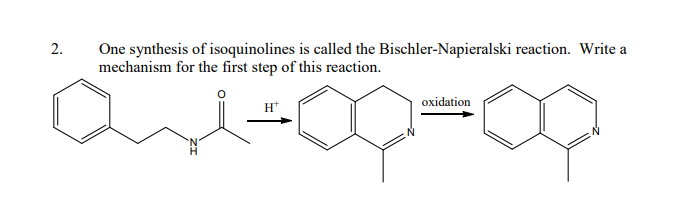
15.98 Describe the Bischler-Napieralski synthesis of iso-quinoline derivative. Give the accepted mechanism of thereactiea. | Snapsolve

Collective Total Synthesis of Aspidofractinine Alkaloids through the Development of a Bischler–Napieralski/Semipinacol Rearrangement Reaction - Wang - 2020 - Angewandte Chemie International Edition - Wiley Online Library

Bischler-Napieralski reaction made by Roman A. Valiulin with ChemDraw | Organic chemistry, Study chemistry, Chemistry education

SciELO - Brasil - The asymmetric total synthesis of (+)- and (-)-trypargine via Noyori asymmetric transfer hydrogenation The asymmetric total synthesis of (+)- and (-)-trypargine via Noyori asymmetric transfer hydrogenation

Bischler Napieralski Reaction Mechanism Application/ Synthesis of Isoquinoline IIT JAM CSIR NET GATE | Chemistry, Organic chemistry, Synthesis

Scheme 2: Noscapine synthesis involving the use of Bischler-Napieralski... | Download Scientific Diagram