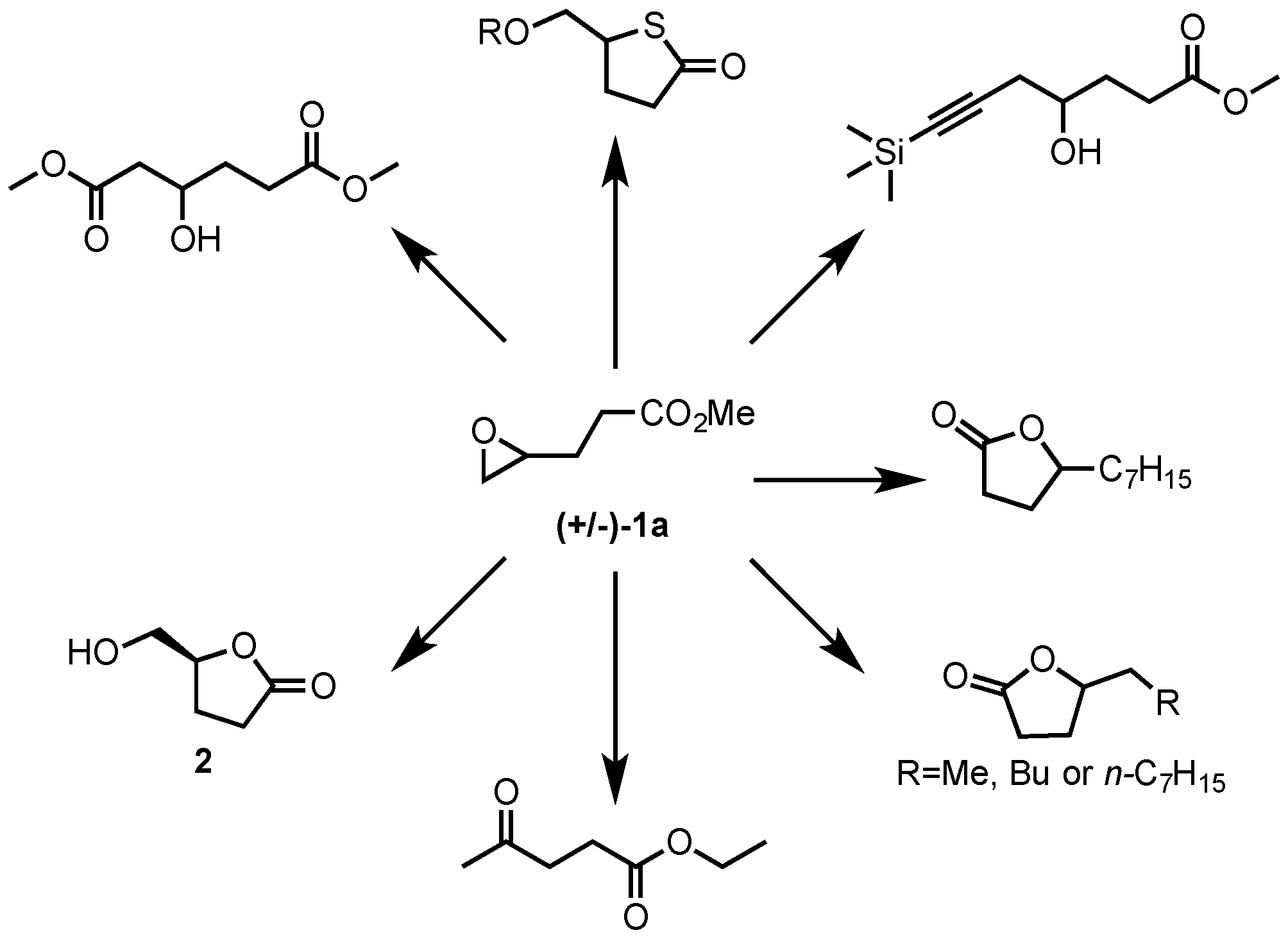
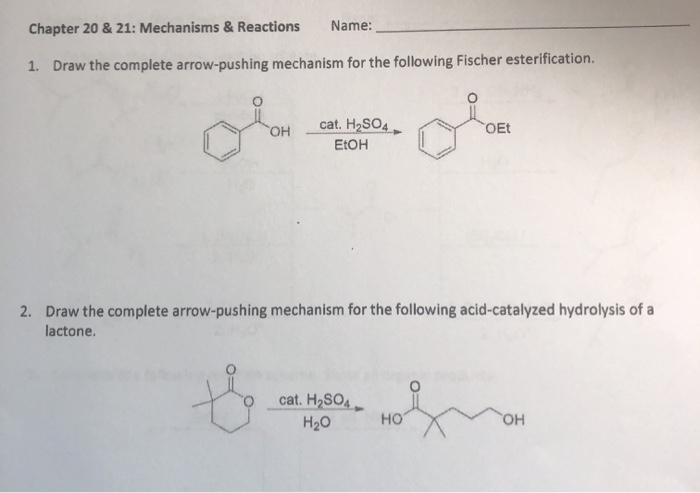
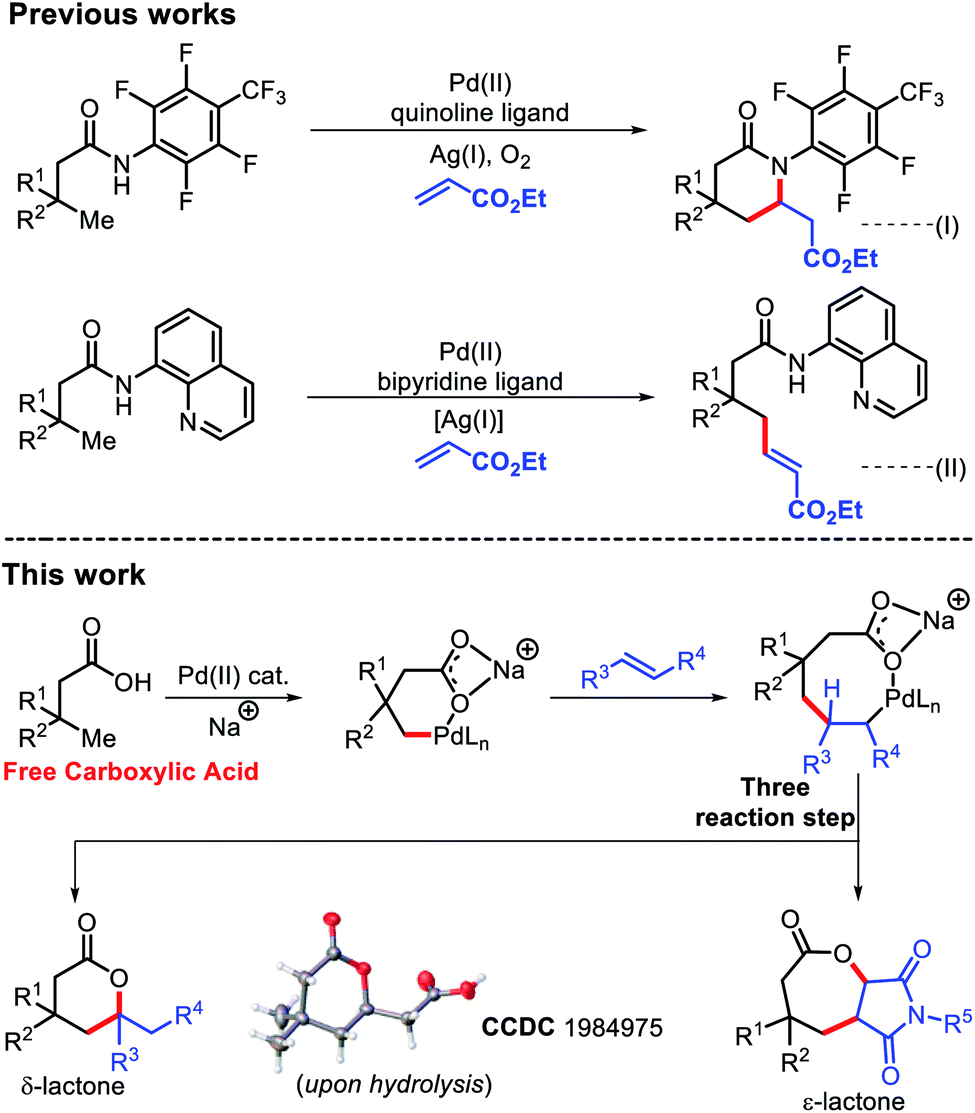
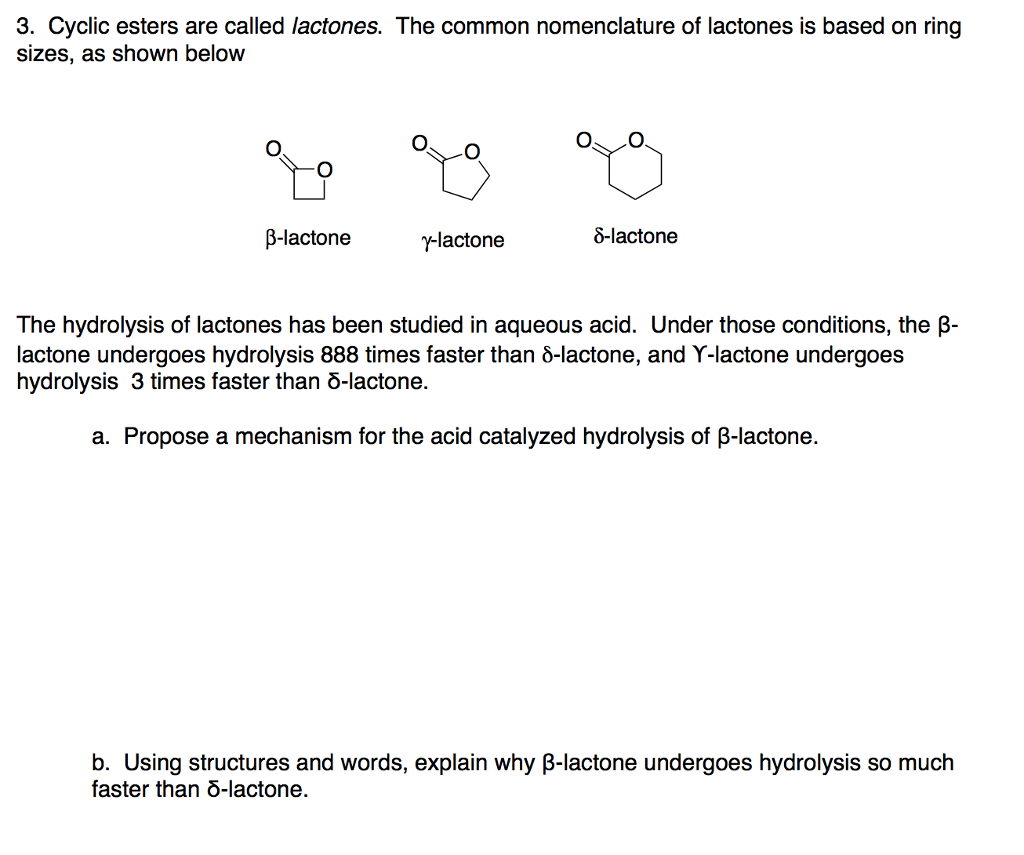
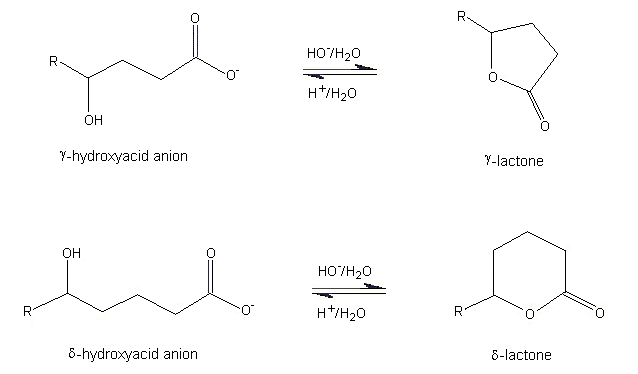
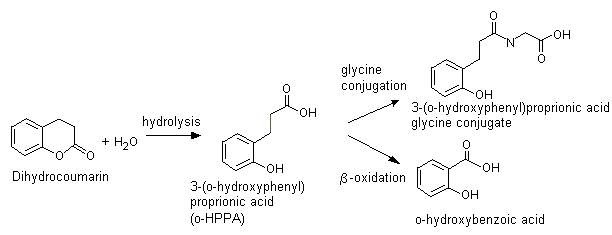
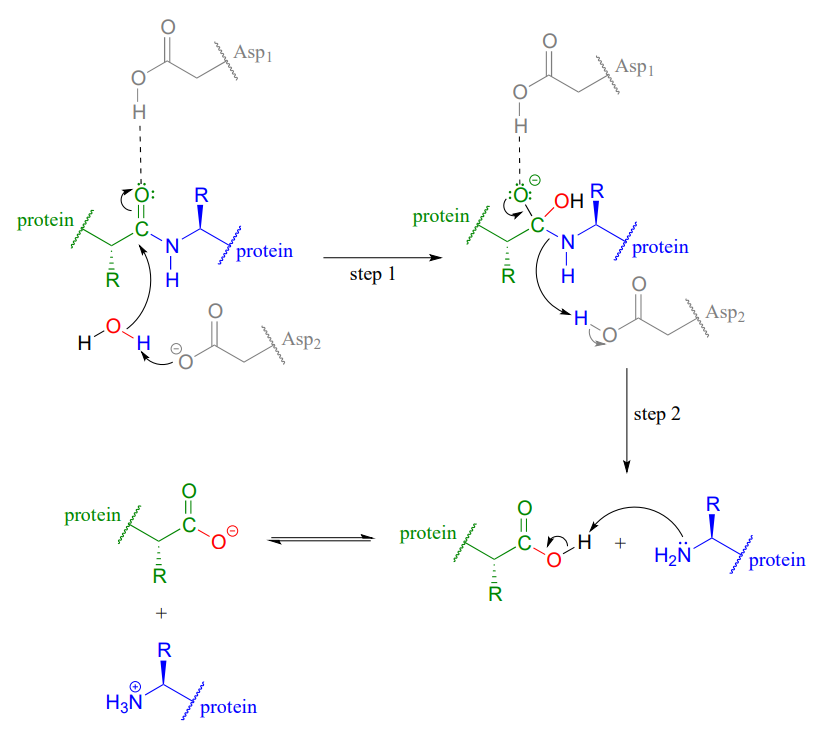
Why does ring opening reaction (of lactones) often follow nucleophilic pathway and not electrophilic?
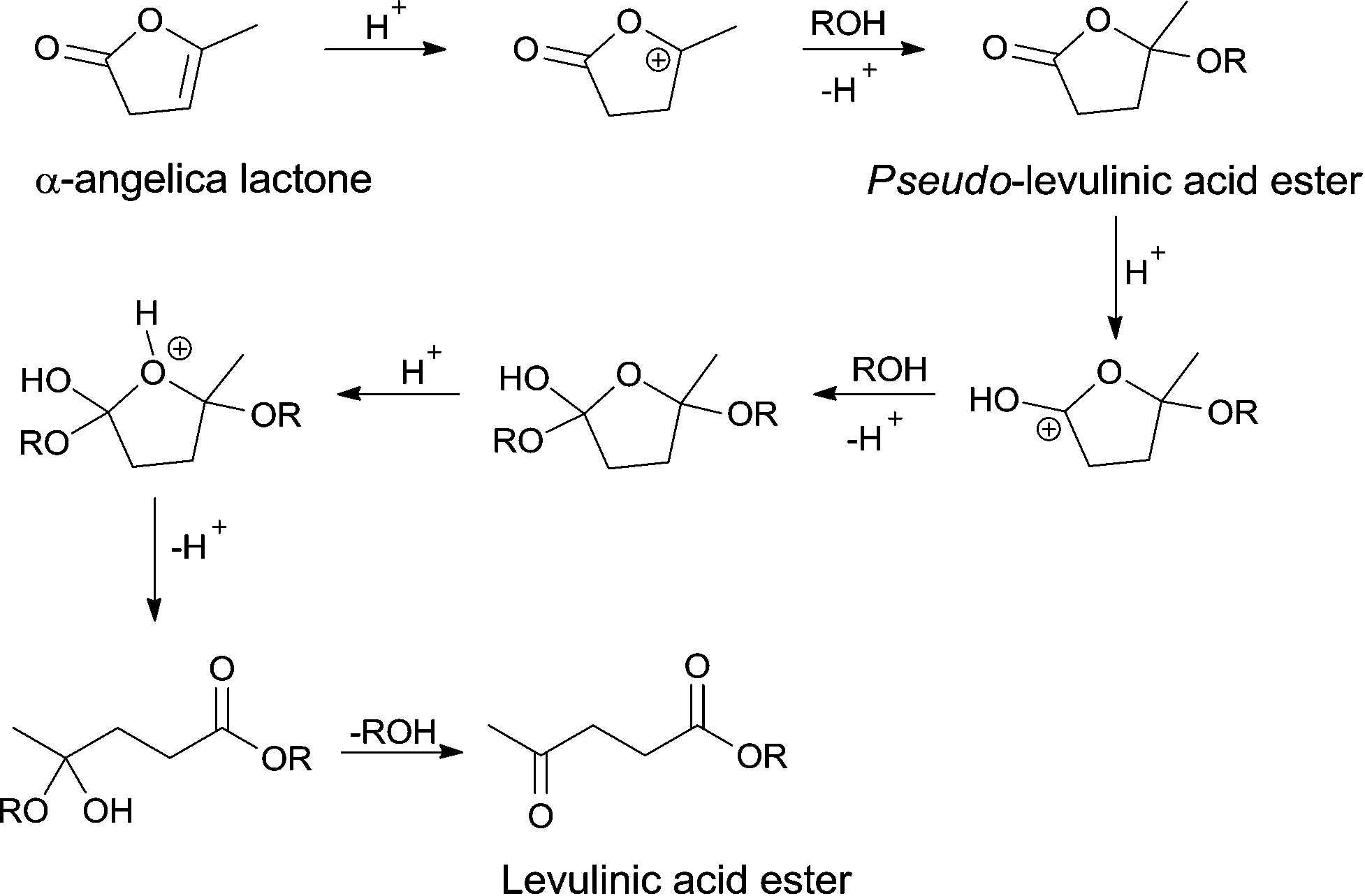
Catalytic upgrading of α-angelica lactone to levulinic acid esters under mild conditions over heterogeneous catalysts - Catalysis Science & Technology (RSC Publishing) DOI:10.1039/C5CY00446B

Table 4 from Mechanisms of lactone hydrolysis in neutral and alkaline conditions. | Semantic Scholar

Mechanisms of Lactone Hydrolysis in Neutral and Alkaline Conditions | The Journal of Organic Chemistry
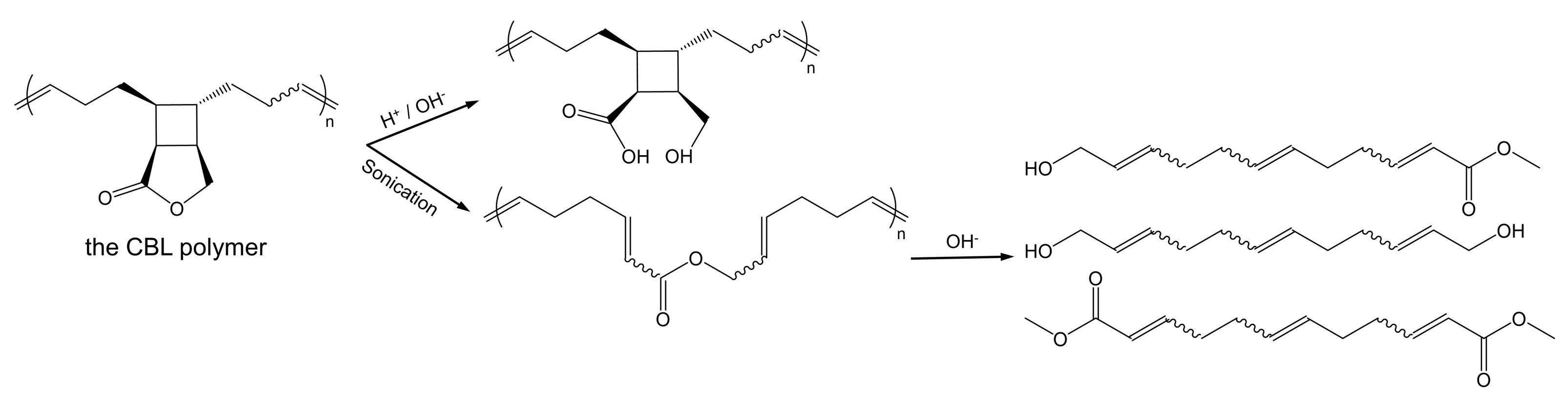
Molecules | Free Full-Text | Mechanisms of a Cyclobutane-Fused Lactone Hydrolysis in Alkaline and Acidic Conditions
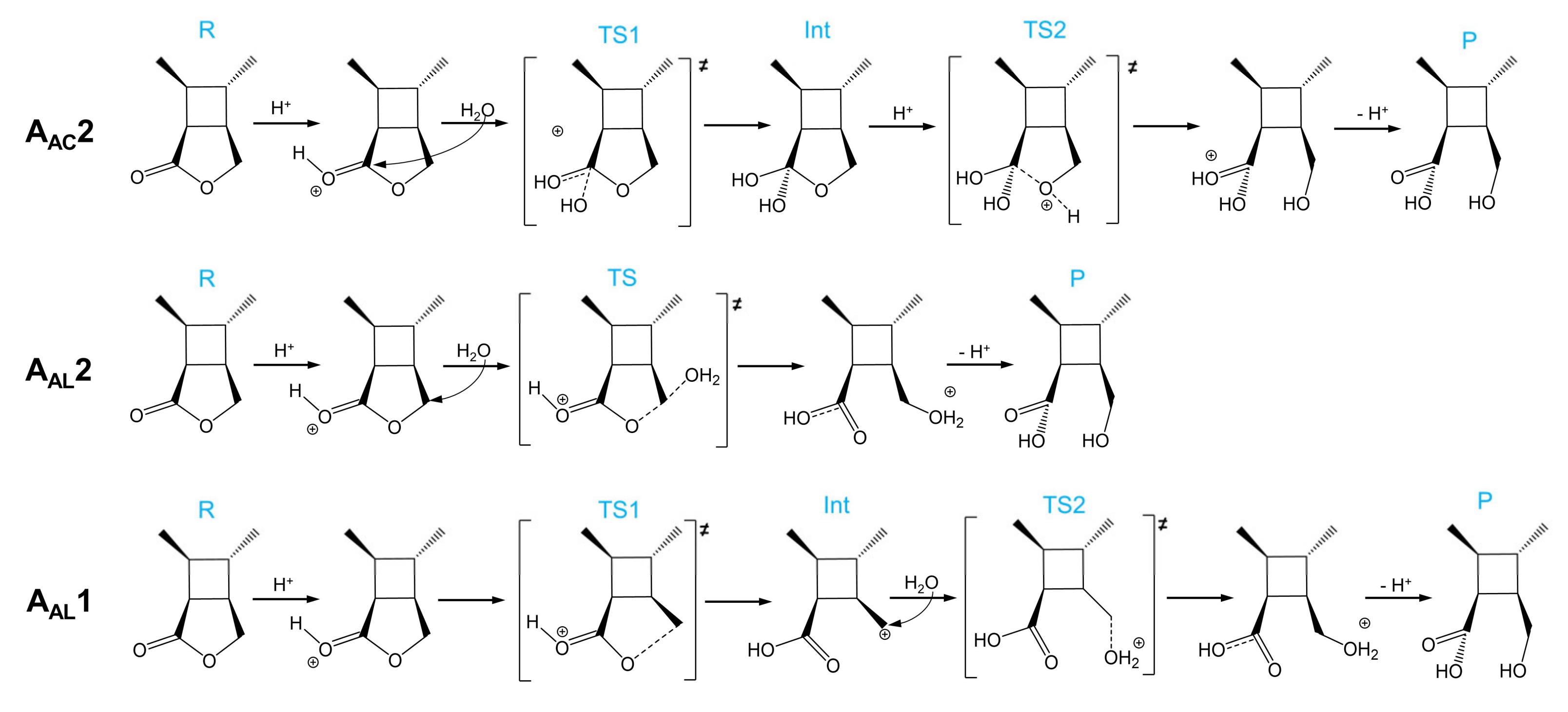
Molecules | Free Full-Text | Mechanisms of a Cyclobutane-Fused Lactone Hydrolysis in Alkaline and Acidic Conditions

Why does ring opening reaction (of lactones) often follow nucleophilic pathway and not electrophilic?

Mechanisms of Lactone Hydrolysis in Neutral and Alkaline Conditions | The Journal of Organic Chemistry