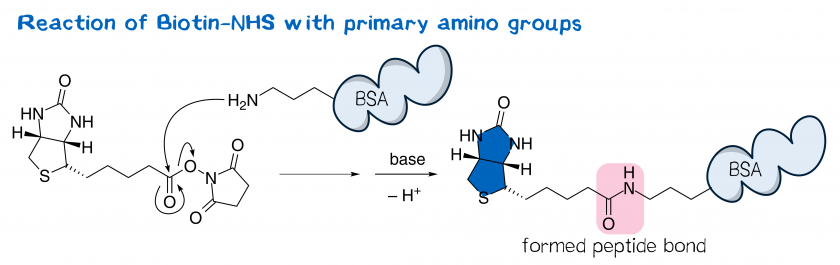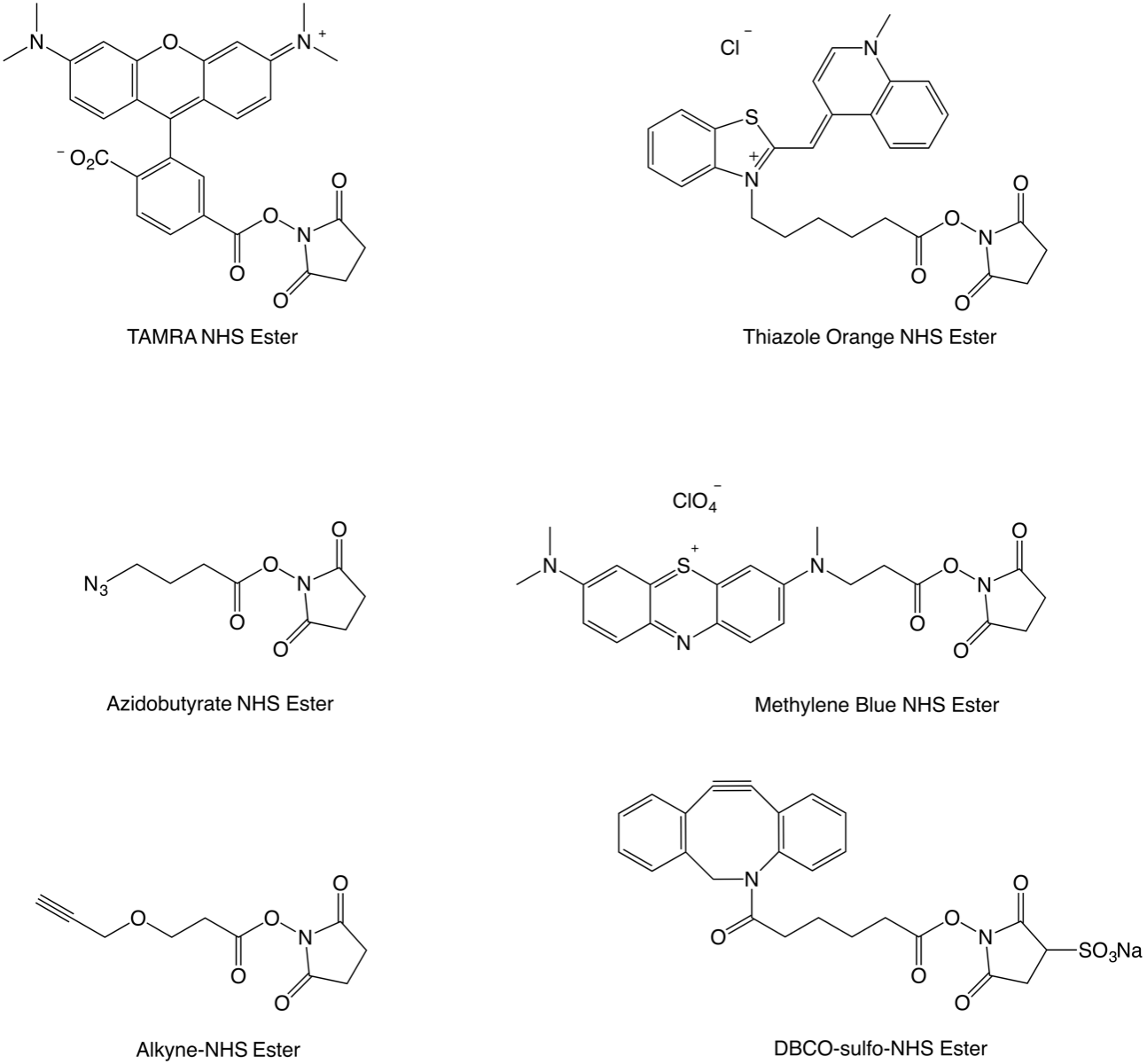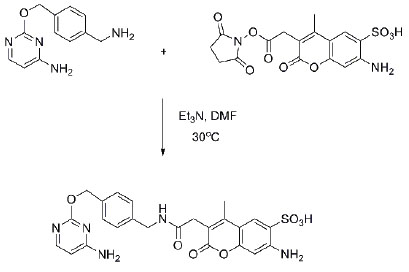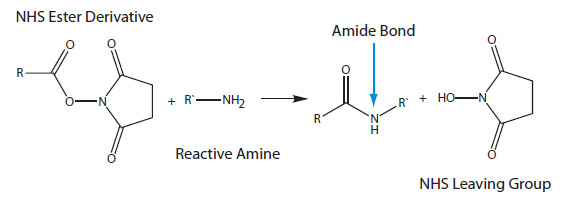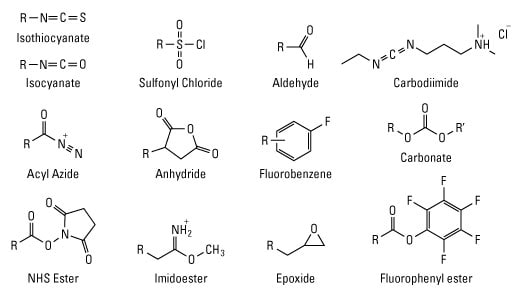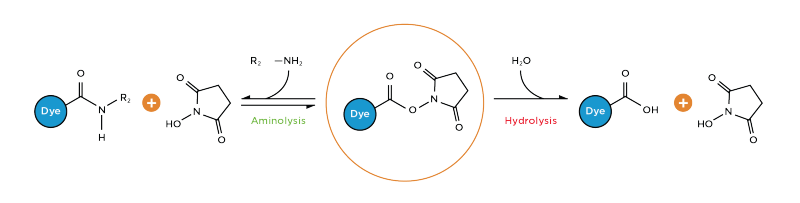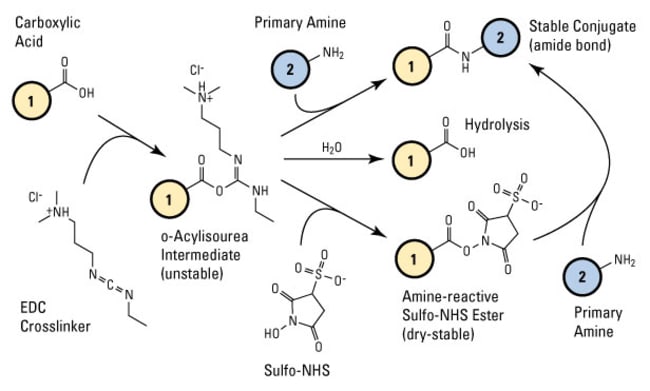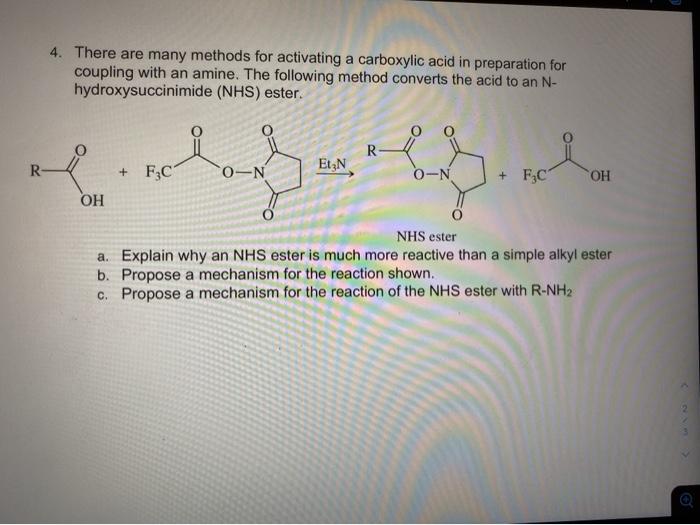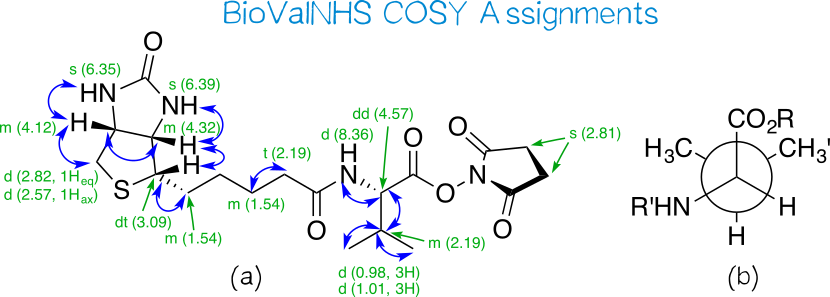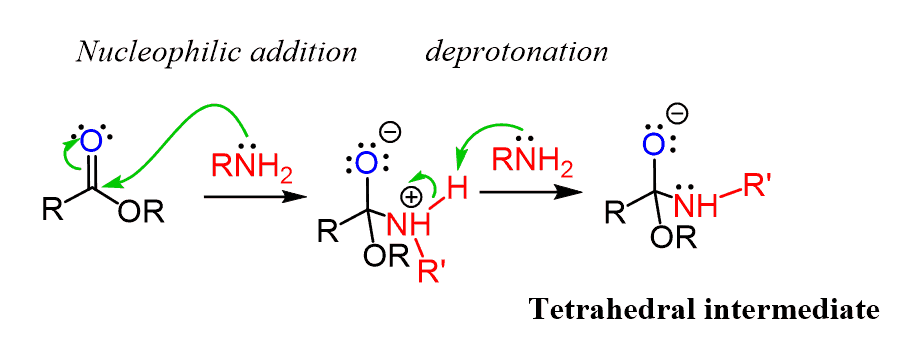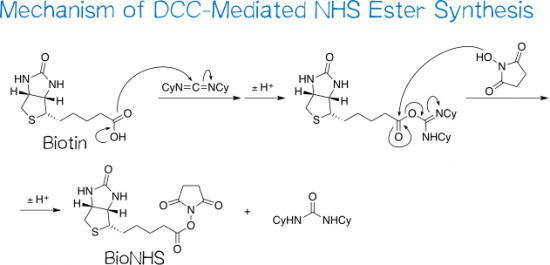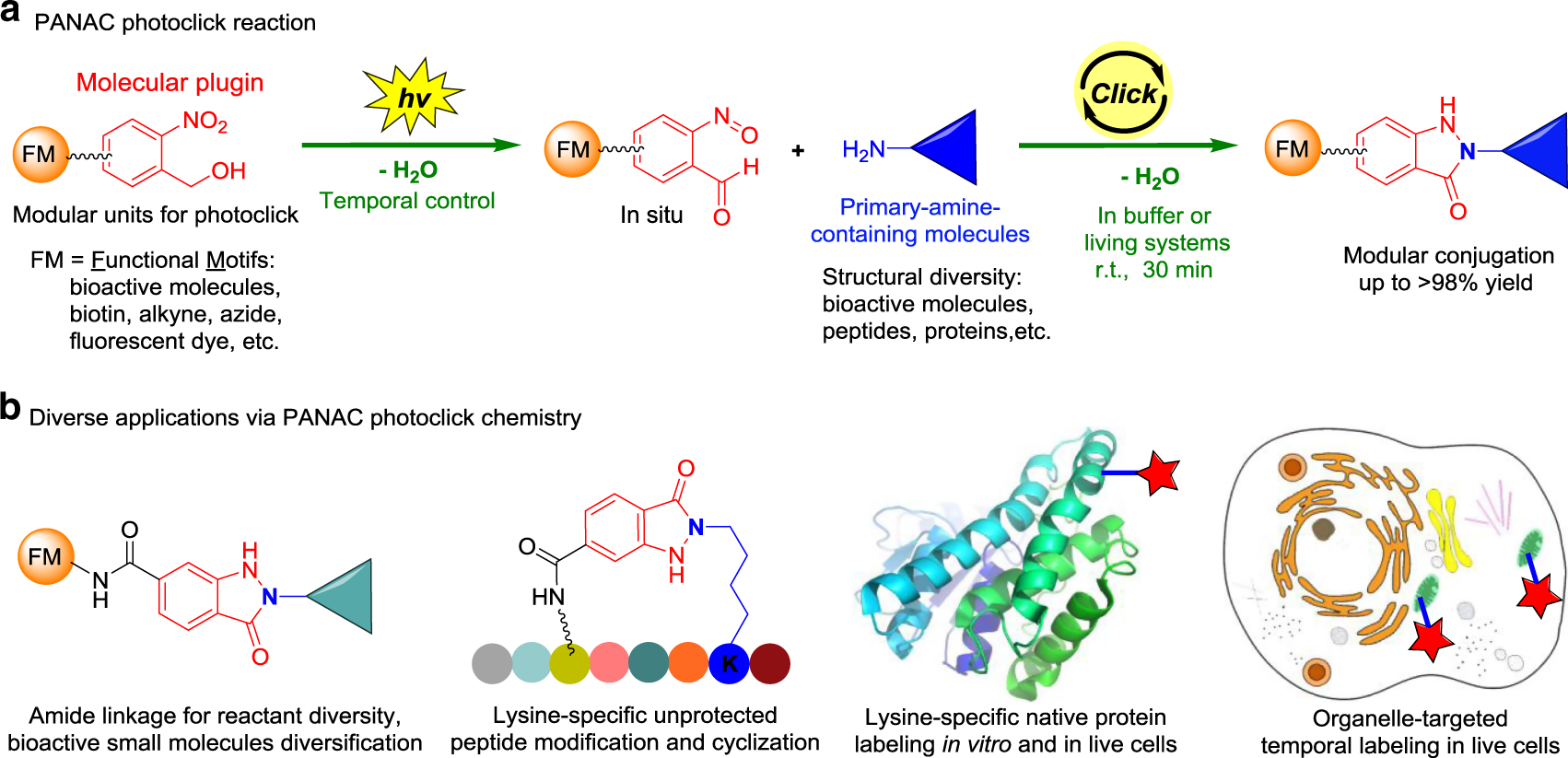
Light-induced primary amines and o-nitrobenzyl alcohols cyclization as a versatile photoclick reaction for modular conjugation | Nature Communications

N-Hydroxysuccinimide-activated esters as a functionalization agent for amino cellulose: synthesis and solid-state NMR characterization | SpringerLink
A method of crosslinking collagen-based material and bioprosthetic devices produced therefrom - Patent 0897942

Amine Analysis Using AlexaFluor 488 Succinimidyl Ester and Capillary Electrophoresis with Laser-Induced Fluorescence
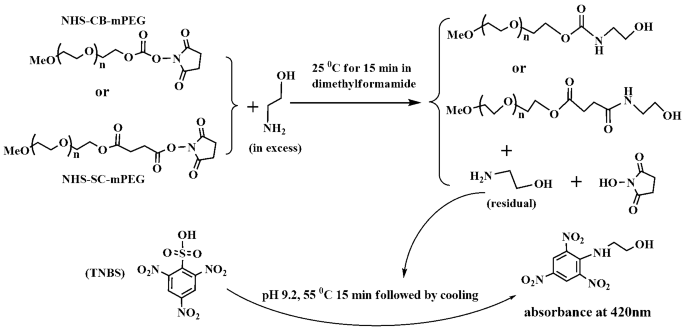
Facile spectrophotometric assay of molar equivalents of N-hydroxysuccinimide esters of monomethoxyl poly-(ethylene glycol) derivatives | BMC Chemistry | Full Text

EDC–NHS chemistry: EDC reacts with a carboxylic-acid group on FEP sheet... | Download Scientific Diagram
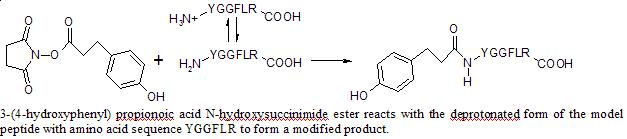
Studying pH Dependence of a Peptide Modification with an N-hydroxysuccinimide Ester Using Mass Spectrometry — Journal of Young Investigators