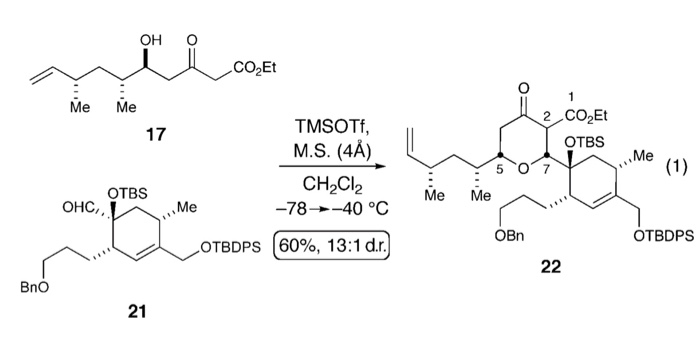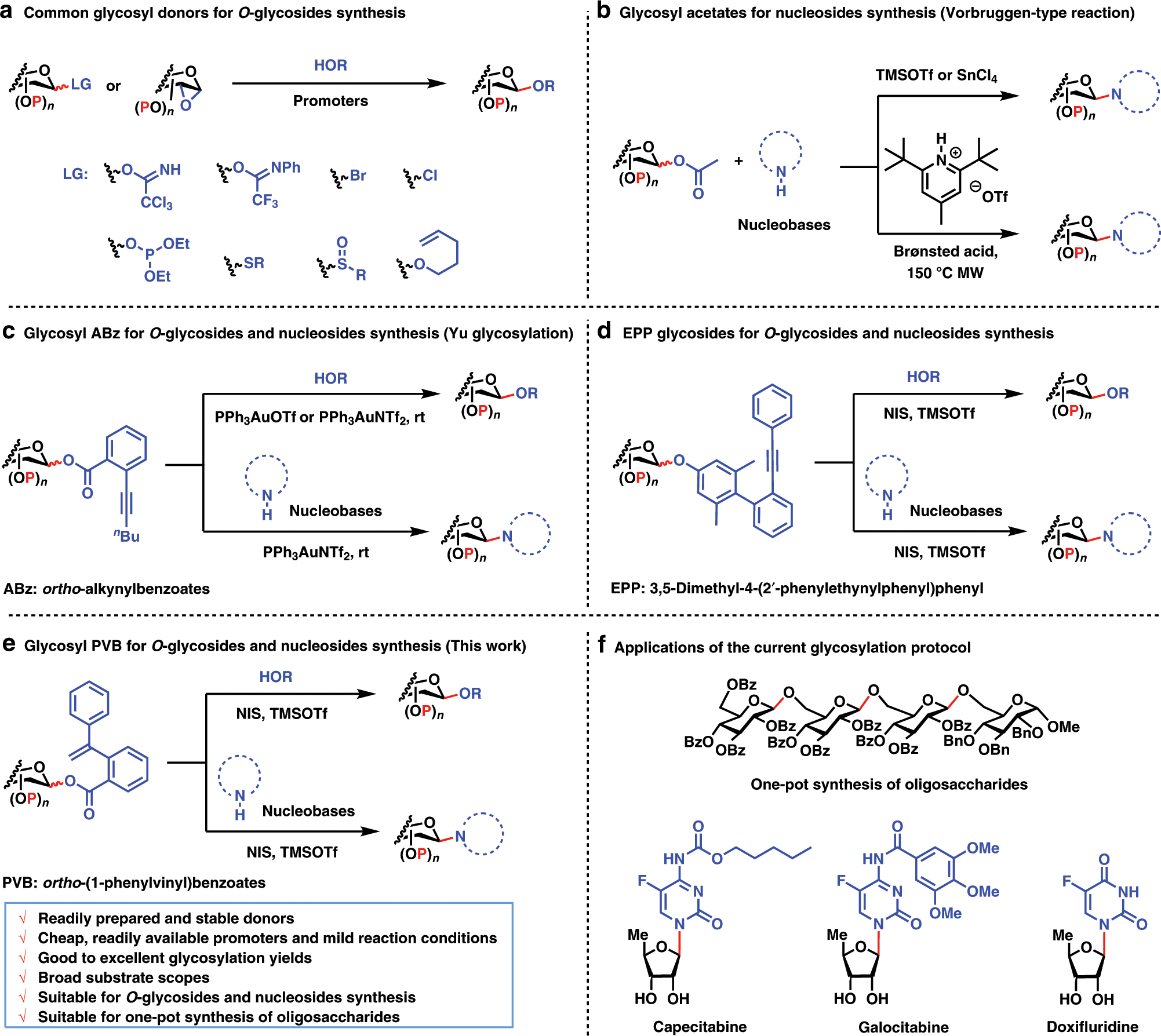
Glycosyl ortho-(1-phenylvinyl)benzoates versatile glycosyl donors for highly efficient synthesis of both O-glycosides and nucleosides | Nature Communications

Figure 1 from Bi(OTf)3-, TfOH-, and TMSOTf-mediated, one-pot epoxide rearrangement, addition, and intramolecular silyl-modified Sakurai (ISMS) cascade toward dihydropyrans: comparison of catalysts and role of Bi(OTf)3. | Semantic Scholar

O‐Trifluoromethylation of N,N‐Disubstituted Hydroxylamines with Hypervalent Iodine Reagents - Matoušek - 2014 - European Journal of Organic Chemistry - Wiley Online Library

Tentative mechanism for the TMSOTf-mediated formation of dithioacetal... | Download Scientific Diagram

Tentative mechanism for the TMSOTf-mediated formation of dithioacetal... | Download Scientific Diagram

TMSOTf‐Catalyzed Silylation: Streamlined Regioselective One‐Pot Protection and Acetylation of Carbohydrates - Joseph - 2012 - European Journal of Organic Chemistry - Wiley Online Library

BF 3 ·OEt 2 and TMSOTf : A synergistic combination of Lewis acids - Chemical Communications (RSC Publishing) DOI:10.1039/B611333H
![Silicon Lewis Acid Catalyzed [3+2] Cycloaddition Reactions of Hydrazones/Cyclopentadiene: Mild Access to Pyrazolidine Derivatives - Zamfir - 2011 - European Journal of Organic Chemistry - Wiley Online Library Silicon Lewis Acid Catalyzed [3+2] Cycloaddition Reactions of Hydrazones/Cyclopentadiene: Mild Access to Pyrazolidine Derivatives - Zamfir - 2011 - European Journal of Organic Chemistry - Wiley Online Library](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/6769206d-9853-43ac-a560-91109269219c/mfig000.jpg)
Silicon Lewis Acid Catalyzed [3+2] Cycloaddition Reactions of Hydrazones/Cyclopentadiene: Mild Access to Pyrazolidine Derivatives - Zamfir - 2011 - European Journal of Organic Chemistry - Wiley Online Library
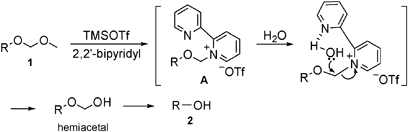
Remarkable effect of 2,2′-bipyridyl : mild and highly chemoselective deprotection of methoxymethyl (MOM) ethers in combination with TMSOTf (TESOTf)–2, ... - Chemical Communications (RSC Publishing) DOI:10.1039/B907910F
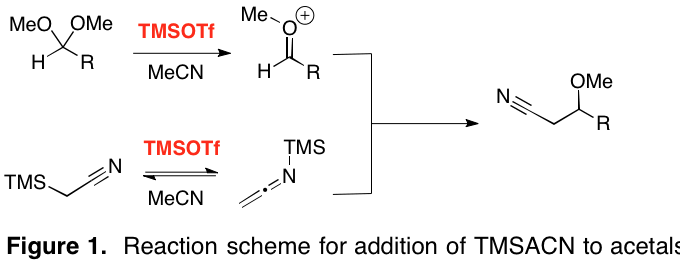
Report: New Reactivity Mediated by Trimethylsilyl Trifluoromethanesulfonate (62nd Annual Report on Research Under Sponsorship of The American Chemical Society Petroleum Research Fund)

The reaction of acetal-type protective groups in combination with TMSOTf and 2,2′-bipyridyl; mild and chemoselective deprotection and direct conversion to other protective groups - ScienceDirect

Scheme 3. Total synthesis of mycalisine B. Reagents and conditions: (a)... | Download Scientific Diagram

Tentative mechanism for the TMSOTf-mediated formation of dithioacetal... | Download Scientific Diagram
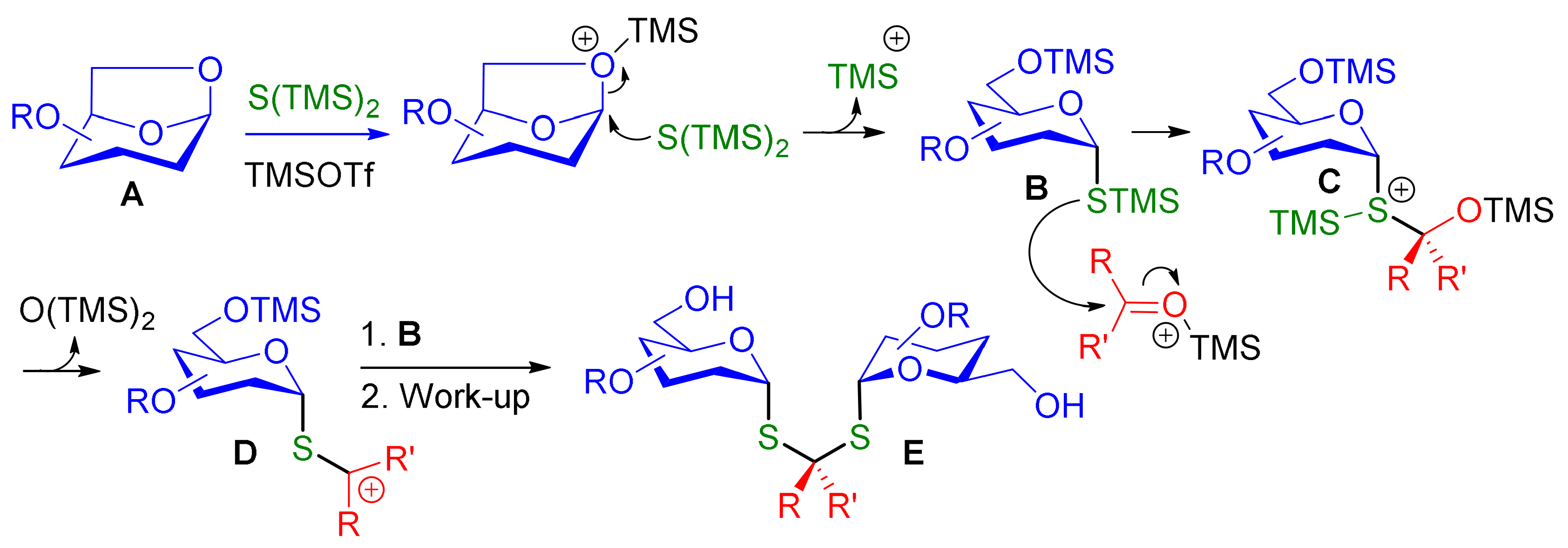
Molecules | Free Full-Text | One-Pot, Highly Stereoselective Synthesis of Dithioacetal-α,α-Diglycosides | HTML

TMSOTf mediated stereoselective synthesis of α-C-glycosides from unactivated aryl acetylenes | SpringerLink
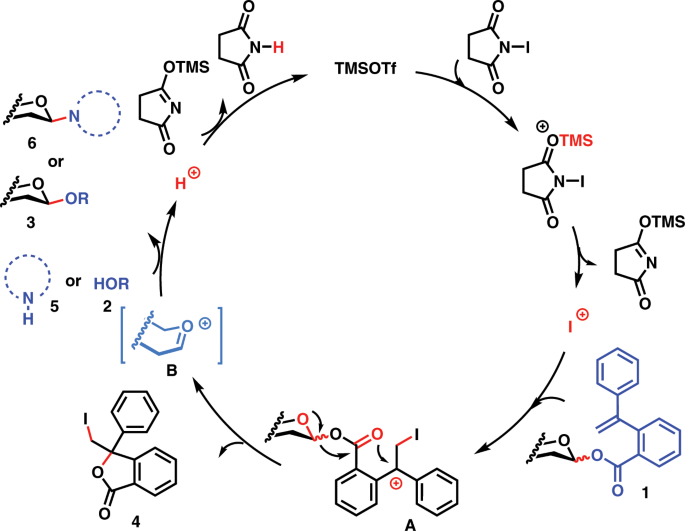
Glycosyl ortho-(1-phenylvinyl)benzoates versatile glycosyl donors for highly efficient synthesis of both O-glycosides and nucleosides | Nature Communications

BF 3 ·OEt 2 and TMSOTf : A synergistic combination of Lewis acids - Chemical Communications (RSC Publishing) DOI:10.1039/B611333H

Scheme 3 Proposed mechanism. Reagents and conditions: (i): 3 or 4 with... | Download Scientific Diagram

Synthesis of 2-O-benzyloxycarbonyl protected thioglucosides (TMSOTf =... | Download Scientific Diagram