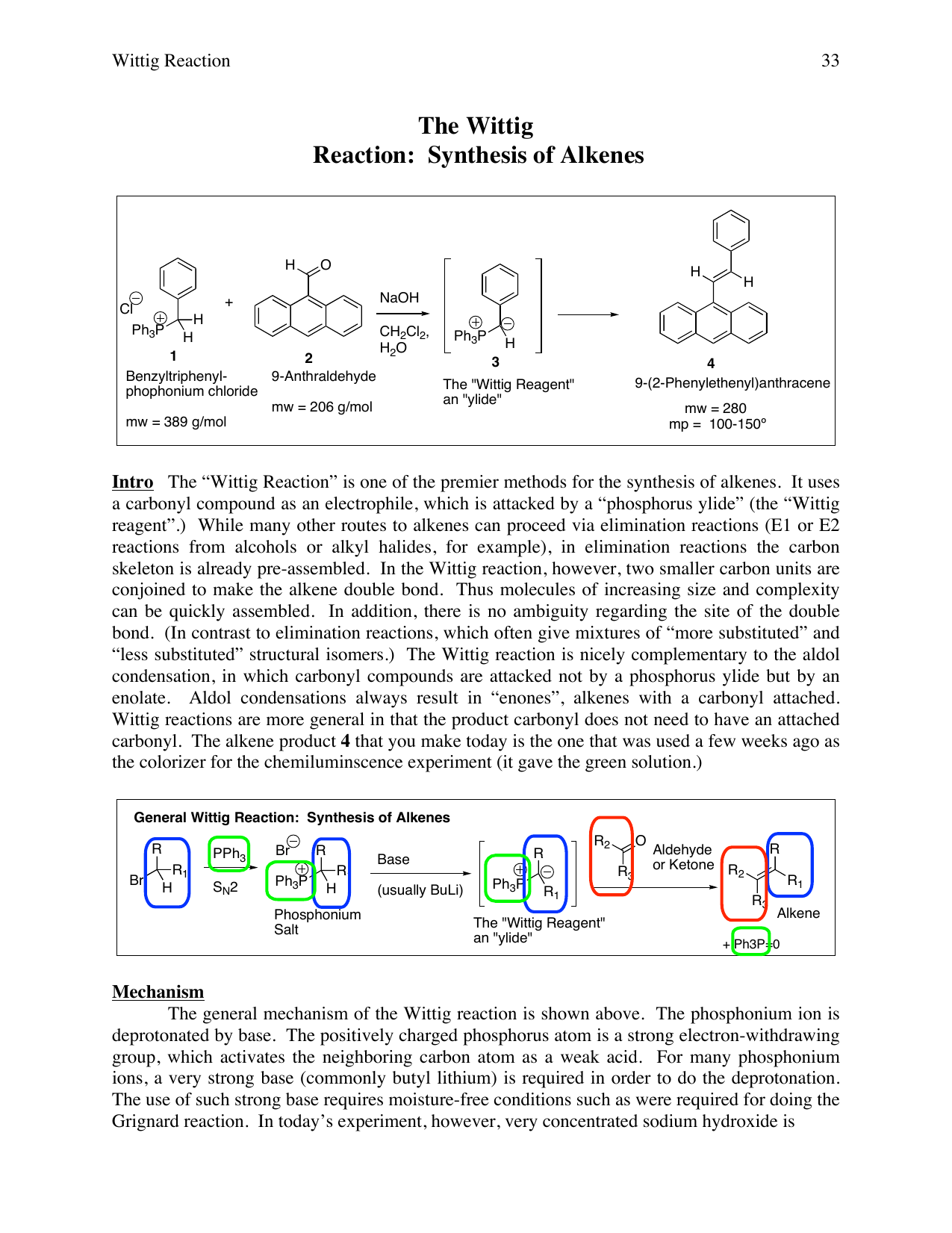
Applications of the Wittig Reaction on the Synthesis of Natural and Natural‐Analogue Heterocyclic Compounds - Rocha - 2018 - European Journal of Organic Chemistry - Wiley Online Library

A Wittig reaction occurs when 4-methylbenzaldehyde and benzyltriphenylphosphonium chloride are stirred together at room temperature in the presence of sodium hydroxide base. Draw the major isomer prod | Study.com

A Stereospecific Wittig Reaction - A STEREOSPECIFIC WITTIG REACTION Kushal Nellore 3/05/ Section: - StuDocu

Wittig Reaction Experiment Part 2: Reaction and Product Isolation - benzyltriphenylphosphonium chloride

A Wittig reaction occurs when 4-methylbenzaldehyde and benzyltriphenylphosphonium chloride are stirred together at room temperature in the presence of sodium hydroxide base. Draw the major isomer prod | Study.com
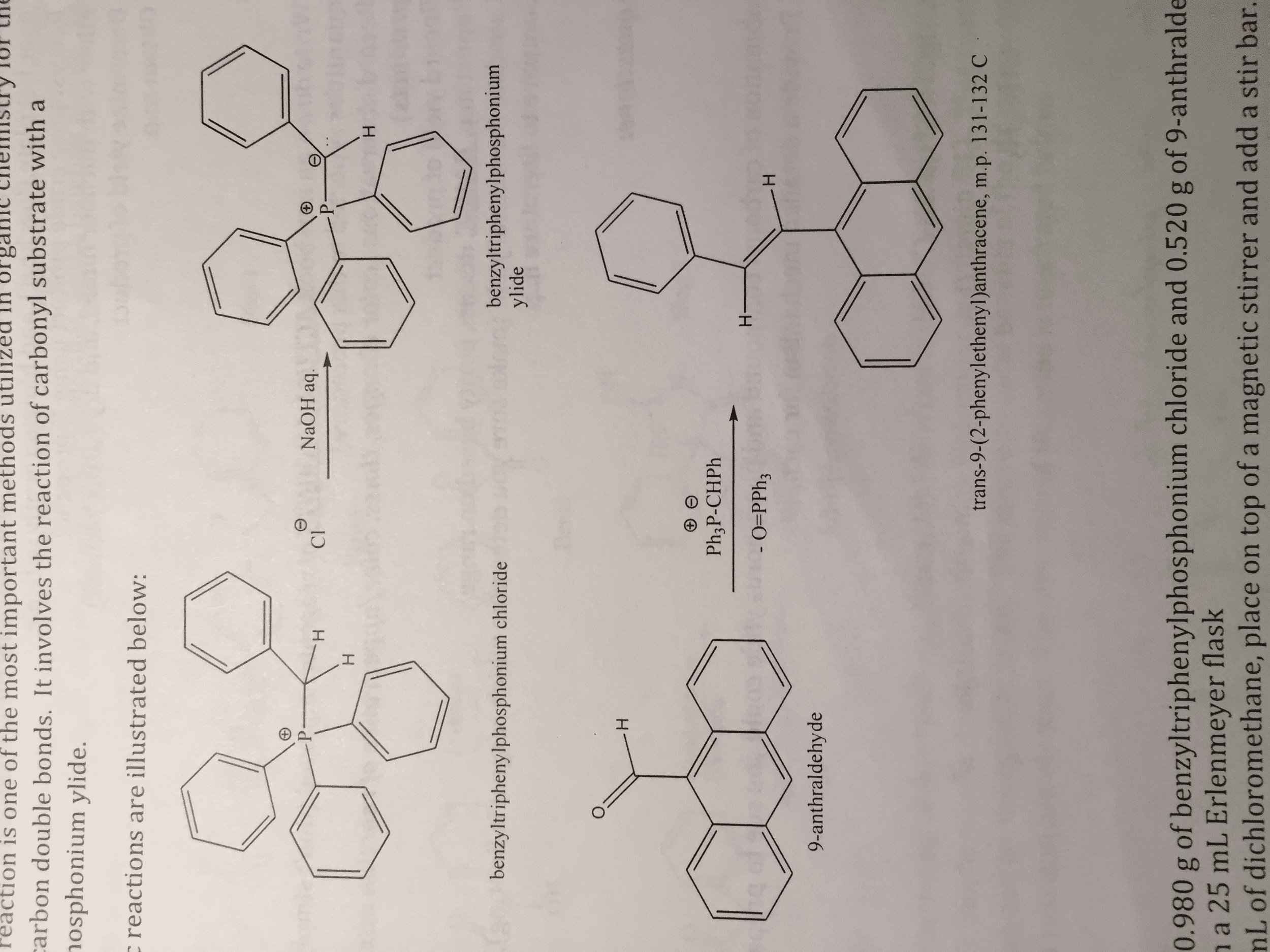
OneClass: What is the mechanism for the synthesis of trans-9(2-phenylethenyl)-anthracene: A Wittig Re...
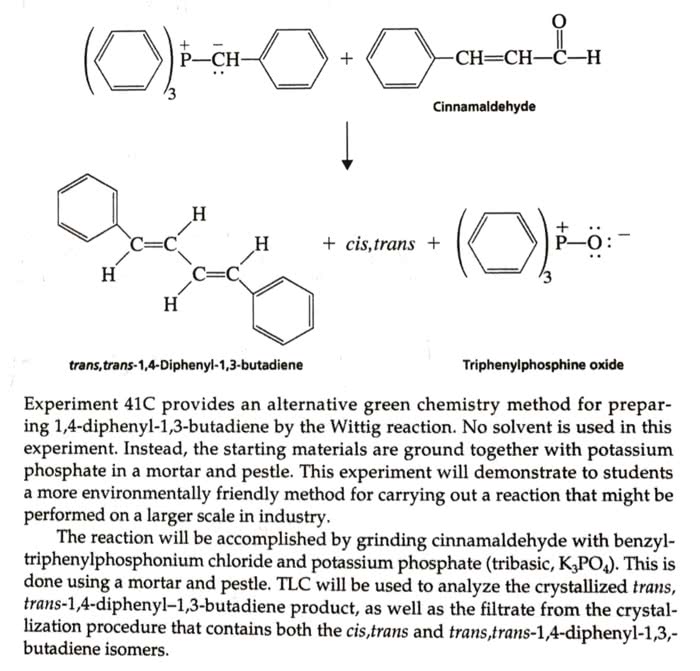
OneClass: Give the balanced equation, theoretical yield, and mechanism for the following experiment i...

What would be the products (if any) of the Wittig reaction using the starting materials below with and the same reagents and conditions as in your experiment (50% NaOH solution and propanol

SOLVED:Wittig Reaction: Complete the reaction of Benzyltriphenylphosphonium Chloride with 50% NaOH and 9-Anthraldehyde. Fisher Esterification: Show the syntheses of n-butylacetate from acetic acid and n- butanol. a) What did we do t0

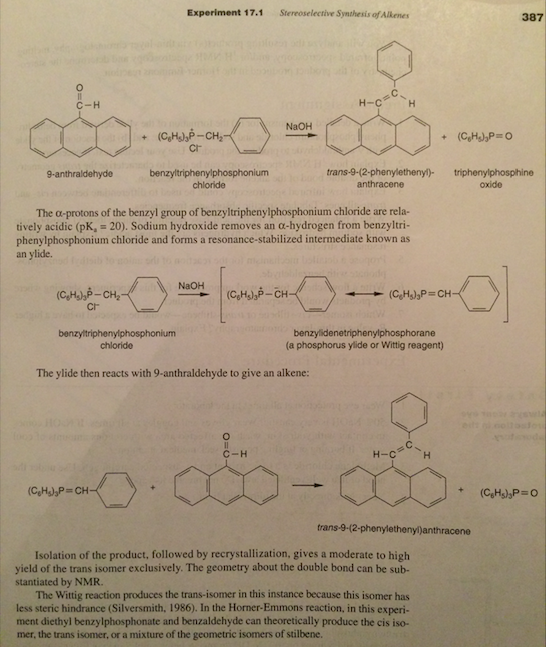
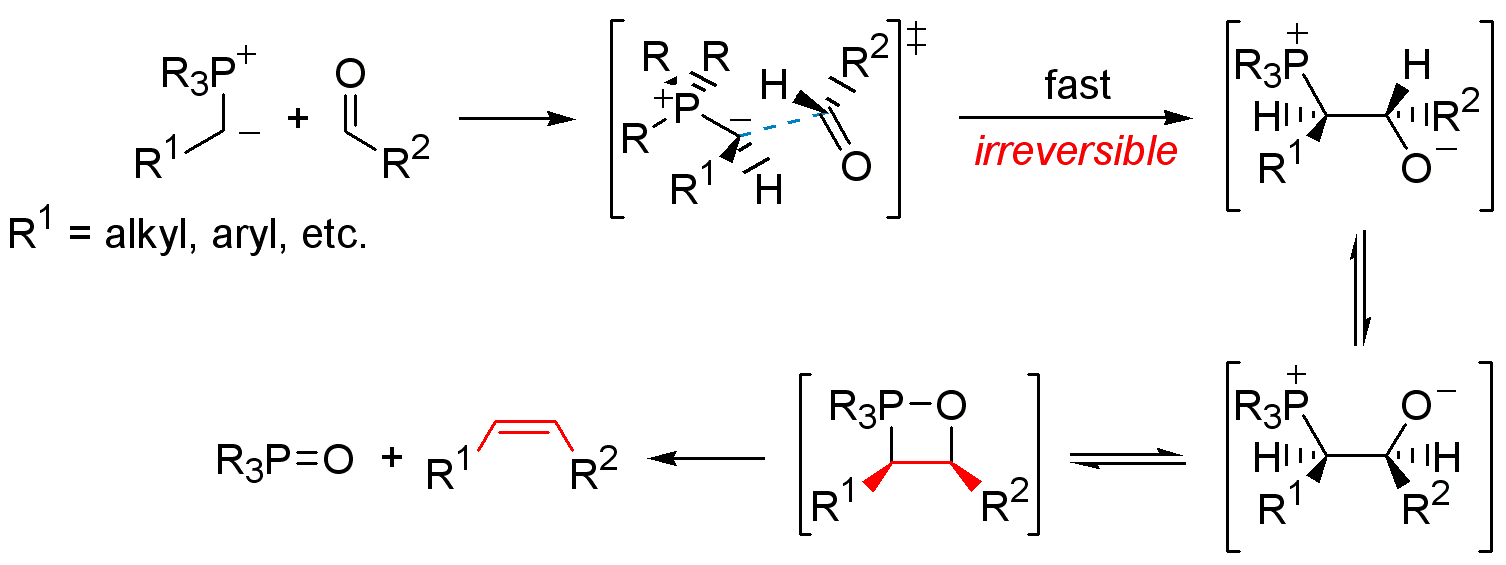
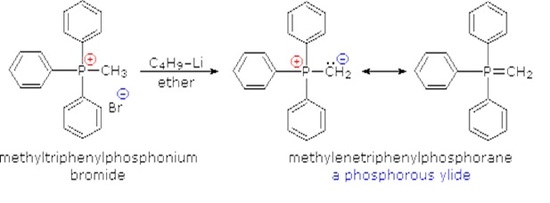


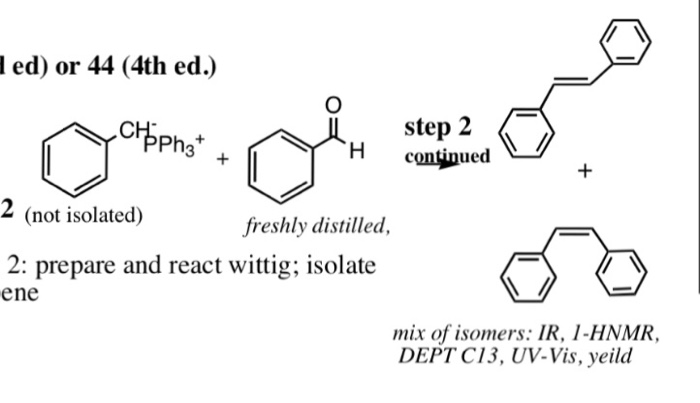
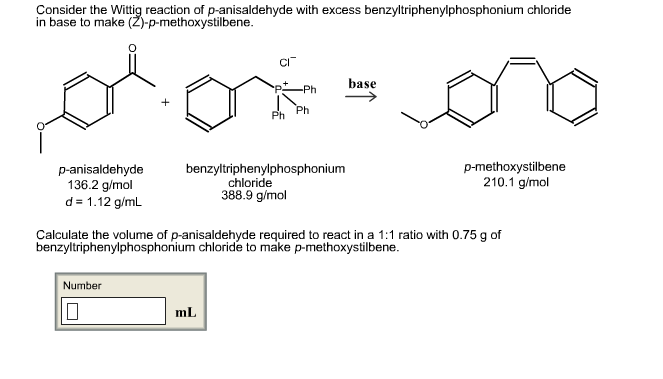
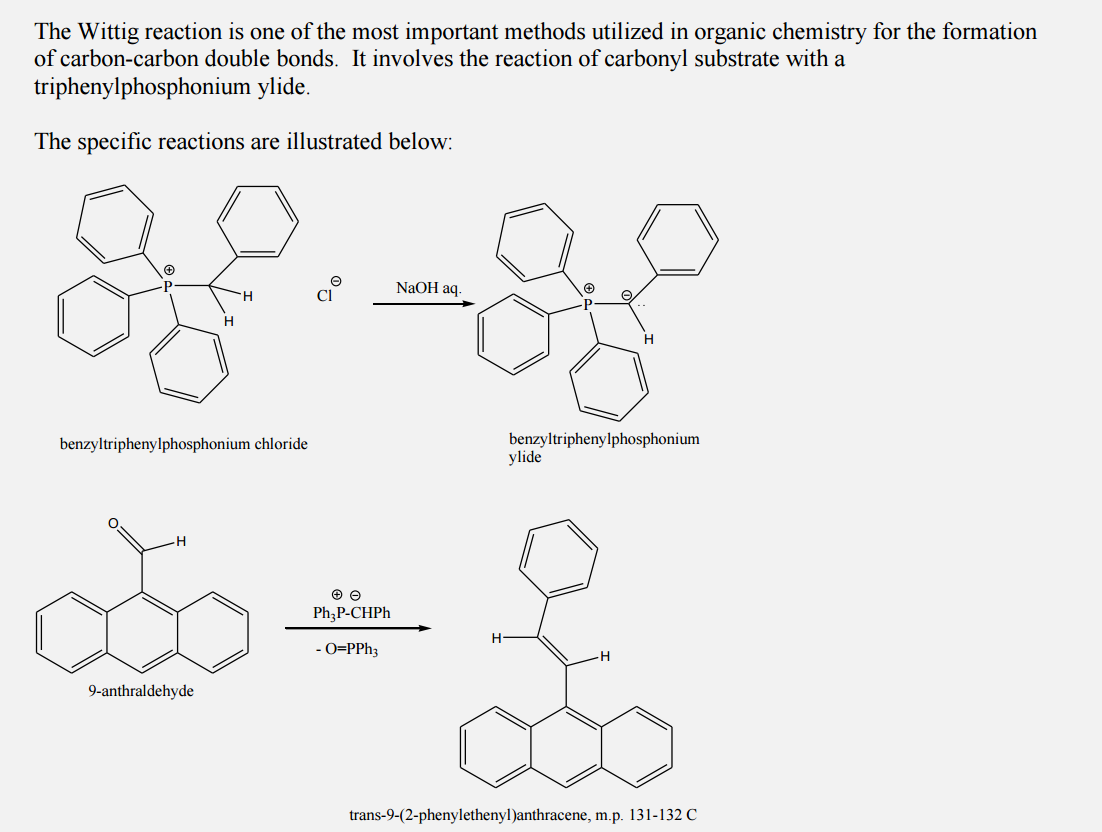
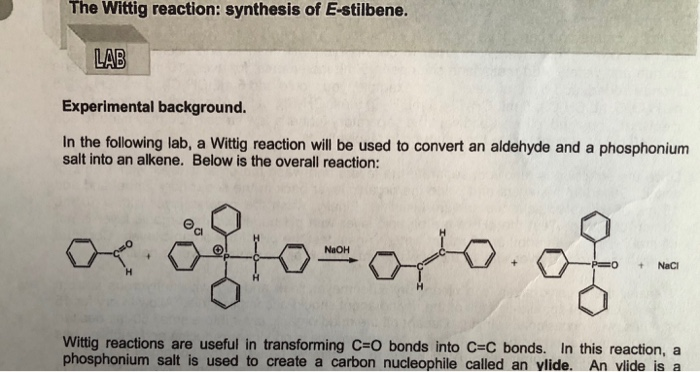


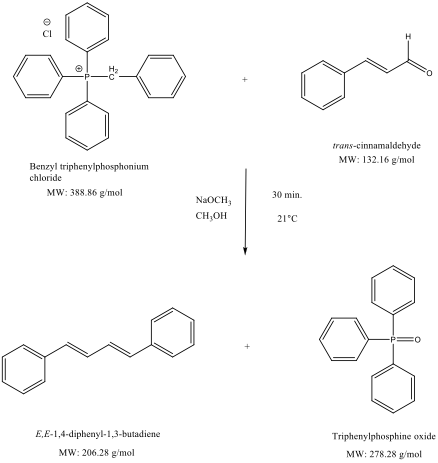
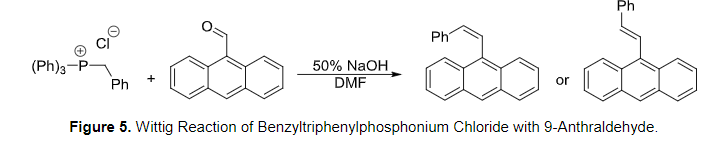


-,%20chloride.jpg)